21st Austria weekly - Valneva (07/01/2020)
12.01.2020, 1019 Zeichen
Valneva: Austrian Frensch biotech company Valneva SE announced that an End Of Phase 2 (EOP2) meeting has been scheduled with the U.S. Food and Drug Administration (FDA) on February 24, 2020 for its single-shot chikungunya vaccine candidate, VLA1553. The Company plans to present its plan for Phase 3 clinical studies and licensure. VLA1553 has been awarded Fast Track designation by the FDA and may be eligible for a Priority Review Voucher. Wolfgang Bender, chief medical officer of Valneva, commented: “We are extremely pleased that the FDA has accepted our proposal for an EOP2 meeting. Subject to the outcome we may commence Phase 3 clinical trials within a few months. We are strongly committed to advancing VLA1553, currently the only chikungunya vaccine candidate in clinical development showing fully sustained titers one year after a single vaccination, through the accelerated approval.”
Valneva: weekly performance:
(From the 21st Austria weekly https://www.boerse-social.com/21staustria (07/01/2020)

Wiener Börse Party #1145: ATX dank Andritz mit Hydropower fester, Raiffeisen Research passt den Game Plan an
Bildnachweis
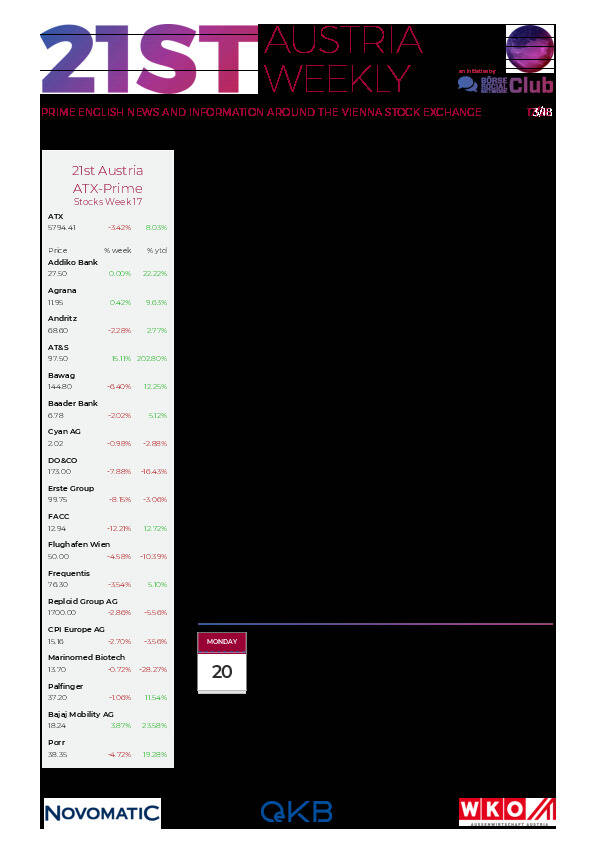
Aktien auf dem Radar:AT&S, UBM, Strabag, Polytec Group, Lenzing, EuroTeleSites AG, Uniqa, ATX, ATX Prime, ATX TR, Wolford, ATX NTR, RBI, OMV, Mayr-Melnhof, Telekom Austria, Palfinger, Andritz, Marinomed Biotech, Österreichische Post, voestalpine, BKS Bank Stamm, Oberbank AG Stamm, Amag, CA Immo, EVN, CPI Europe AG, Kapsch TrafficCom, Semperit, RHI Magnesita.
Random Partner
Bajaj Mobility AG (vormals Pierer Mobility AG)
Die Bajaj Mobility AG (vormals PIERER Mobility AG) ist die Holdinggesellschaft der KTM-Gruppe, einem der führenden Motorradhersteller Europas. Mit ihren Marken KTM, Husqvarna und GASGAS zählt die KTM AG zu den europäischen Premium-Motorradherstellern. Das Produktportfolio umfasst neben Motorrädern mit Verbrennungsmotor auch High-End-Komponenten (WP) sowie Fahrzeuge mit innovativen Elektroantrieben.
>> Besuchen Sie 55 weitere Partner auf boerse-social.com/partner
Useletter
Die Useletter "Morning Xpresso" und "Evening Xtrakt" heben sich deutlich von den gängigen Newslettern ab.
Beispiele ansehen bzw. kostenfrei anmelden. Wichtige Börse-Infos garantiert.
Newsletter abonnieren
Runplugged
Infos über neue Financial Literacy Audio Files für die Runplugged App
(kostenfrei downloaden über http://runplugged.com/spreadit)
per Newsletter erhalten