21st Austria weekly - Marinomed, Valneva (23/09/2021)
26.09.2021, 2239 Zeichen
Marinomed: Marinomed Biotech AG, an Austrian science-based biotech company with globally marketed therapeutics derived from innovative proprietary technology platforms, announced today that starting October 2021, Stephanie Kniep will be heading the company’s Investor Relations (IR) activities. With her vast IR experience and knowledge, she will contribute to Marinomed’s strategic communications and positioning on the international capital markets. Stephanie Kniep is a renowned IR Manager with more than 20 years of experience.
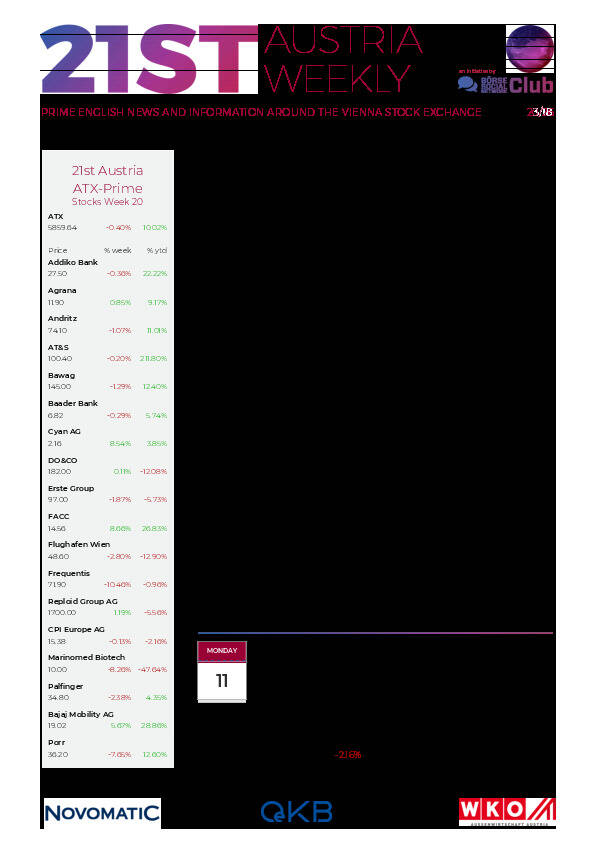
Marinomed Biotech: weekly performance:
Valneva: Valneva, an Austrian/French specialty vaccine company, announced that it has commenced recruitment of adolescents in its pivotal Phase 3 Clinical Trial (VLA2001-301, “Cov-Compare”) for its inactivated COVID-19 vaccine candidate VLA2001 in the United Kingdom. Topline results from the pivotal Cov-Compare trial are expected early in the fourth quarter of 2021 and are intended to form the basis for potential regulatory approval in adults. The Company has also started to provide boosters to volunteers in its Phase 1/2 VLA2001-201 trial. This planned expansion of VLA2001 clinical trials will support future approval in further age groups, in addition to adults. Further, the company said it continues discussions with the European Commission regarding a potential VLA2001 supply contract. The Company is also actively pursuing opportunities to make VLA2001 available to other customers, subject to positive Cov-Compare data and regulatory approval. CEO Thomas Lingelbach commented, “Our teams at Valneva remain fully committed to carry out VLA2001’s development plan and bring our inactivated vaccine to all patient groups who could benefit. We continue to receive messages on a daily basis from people across the world who are waiting for an inactivated vaccine so we continue to believe that our differentiated vaccine candidate could contribute to the ongoing fight against the COVID-19 pandemic. We’re confident that many countries, and regulators, will want to have the opportunity to consider our inactivated COVID-19 vaccine.”
Valneva: weekly performance:
(From the 21st Austria weekly https://www.boerse-social.com/21staustria (23/09/2021)

Wiener Börse Party #1161: ATX nach Wiener Börse Preis mit Sieger VIG (O-Ton Hartwig Löger hier) im 6000er-Flirt; AT&S 10 Prozent Plus
Bildnachweis
Aktien auf dem Radar:AT&S, FACC.
Random Partner
RWT AG
Die Firma RWT Hornegger & Thor GmbH wurde 1999 von den beiden Geschäftsführern Hannes Hornegger und Reinhard Thor gegründet. Seitdem ist das Unternehmen kontinuierlich, auf einen derzeitigen Stand von ca. 30 Mitarbeitern, gewachsen. Das Unternehmen ist in den Bereichen Werkzeugbau, Formenbau, Prototypenbau und Baugruppenfertigung tätig und stellt des Weiteren moderne Motorkomponenten und Präzisionsteile her.
>> Besuchen Sie 55 weitere Partner auf boerse-social.com/partner
Useletter
Die Useletter "Morning Xpresso" und "Evening Xtrakt" heben sich deutlich von den gängigen Newslettern ab.
Beispiele ansehen bzw. kostenfrei anmelden. Wichtige Börse-Infos garantiert.
Newsletter abonnieren
Runplugged
Infos über neue Financial Literacy Audio Files für die Runplugged App
(kostenfrei downloaden über http://runplugged.com/spreadit)
per Newsletter erhalten